What is near-infrared spectroscopy?
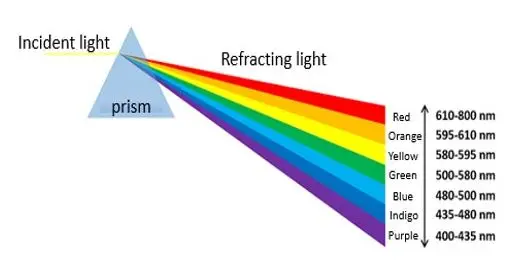
Infrared radiation (IR) was first detected in 1800 by Friedrich Wilhelm Herschel. He wanted to know if there was a specific colour of light that could be related to the warmth of sunlight. Herschel used a prism to separate the colours of light and held a thermometer as a detector to the different rainbow colours or spectral colours.
Beyond the red end of the visible spectrum, he found the maximum amount of heat. Since there was no visible light, Herschel surmised that there must be an invisible form of light beyond the red end of the visible spectrum. This radiation, which was invisible to him, he called infrared radiation (IR).
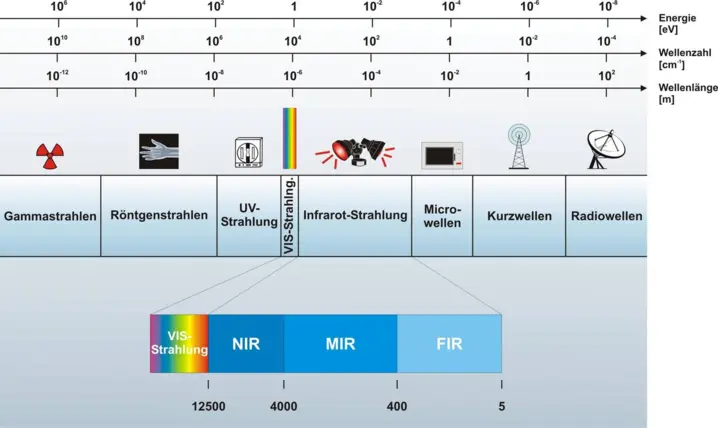
Today we know that infrared radiation can be divided even further. Infrared radiation is composed of several so-called spectral ranges. To make the different spectral ranges usable, different radiation sources and optical components are built into a spectrometer.
One of the most versatile and useful types of IR spectrometers is the near-infrared (NIR) spectrometer, which covers wavelengths just beyond the visible, i.e. from 780 nm to 2500 nm.
The theory of molecular spectroscopy
Spectroscopy is an analytical technique that relies on the interaction of electromagnetic radiation with matter to determine its chemical composition and structure
The acquired data are presented as a spectrum, defined as a graphical representation of the detected intensity of electromagnetic radiation as a function of wavelength
In IR Spectroscopy, molecules absorb infrared radiation at specific frequencies, inducing transitions from the ground state to excited vibrational states. These transitions are associated with vibrational motions of molecular bonds, which primarily occur in two fundamental modes: stretching and bending
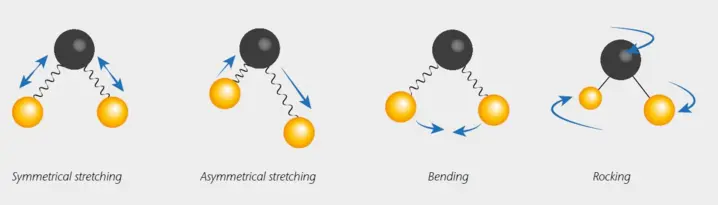
Typically, NIR spectra show broader bands arising from the overlapping harmonics and combination vibrations that occur in more complex molecules. Bonds that can usually be "seen" in NIR spectra are H-C, H-O, H-S and H-N. Organic materials often contain these molecular H-bonds. Due to this fact, biological and organic materials are often analysed with NIR spectroscopy.
General spectrometer design and optical components
The purpose of a spectrometer is to measure the intensity of light (electromagnetic radiation) across different wavelengths, allowing the individual components of the light to be seen and analysed separately. Every spectrometer consists of some key optical components: Light emitted from a source (light source) is first collimated into a parallel beam (collimating optics), then passes through a dispersive element (diffraction grating), and is finally the spectrum is focused by optics onto a detector (photodiode array).
Light source: The light source emits the specific infrared radiation. Different light sources are used for the different spectral ranges. For NIR spectroscopy, a halogen lamp is used.
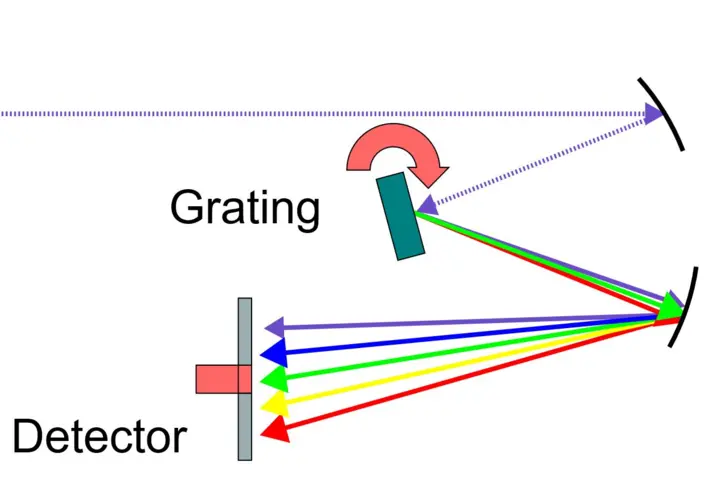
Diffraction element / grating: A type of prism is used to split the light emitted by the light source into the different spectral ranges. The Polytec NIR spectrometers use a grating. A grating is an optical element that diffracts (splits) electromagnetic radiation according to the energy difference. A common type is a reflective surface with a regular array of lines/grooves etched into the surface. The number of lines, profile and depth determine the spectral range, efficiency, resolution and overall performance. For a line grating, lines are often cut into a glass substrate with a reflective coating using a diamond-tipped tool.
The second important type of grating is the holographic volume phase grating. Holographic gratings are produced by a photolithographic process, usually in a matrix material (dichromated gelatin) sandwiched between transparent covers (glass). Diffraction occurs due to a periodic change in the refractive index at constant thickness.
Detector / photodiode array: In the spectrometer, a photodiode array is used as the detector unit to measure the light intensity. A photodiode array is a semiconductor component. The detector element consists of a group of linearly arranged photodiodes as well as a supply and readout circuit. The semiconductor material used for these diodes depends on the spectral sensitivity for the required wavelength range. For the near-infrared spectrum and the spectral range of 900-2500 nm, InGaAs (indium gallium arsenide) or PbS (lead sulphide) is used.
Spectrometer constructions: CzernyTurner (CZ) and transmission spectrograph (TR)
Two very commonly used spectrometer designs are the CzernyTurner spectrograph, which is usually based on a reflective grating and mirrors, and the transmission spectrograph.
The biggest problems with CZ spectrographs are stray light due to grating imperfections and the lower throughput of the system due to the reflective grating and the lower reflectivity of mirrors compared to the transmission of lenses. In addition, mirror-based systems are more difficult to compensate for optical errors such as chromatic aberration or astigmatism.
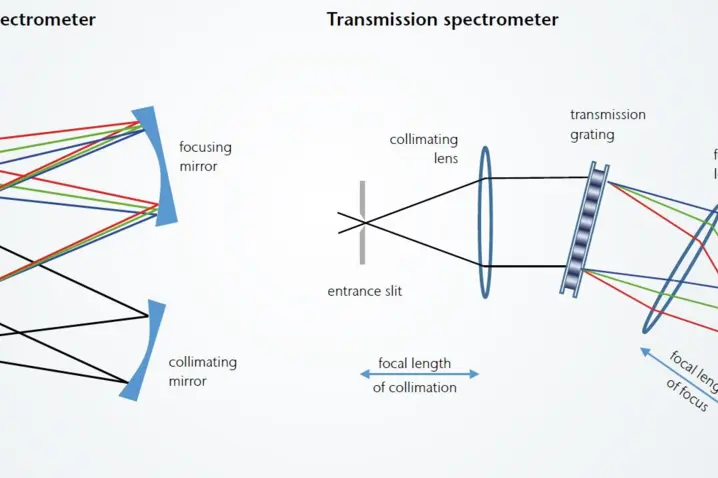
Advantages of transmission design:
The main advantage of a transmission grating-based spectrometer is higher throughput (compared to reflectance spectrometers). The greatest contribution to this is the higher diffraction efficiency of volume phase holographic gratings (VPH). This results from the higher regularity of VPH gratings and the possibility to apply anti-reflective coatings to the covers of VPH gratings. Reflective gratings (and the mirrors used in a CZ spectrograph) have a lower throughput due to surface losses in the reflection process. When designing VPH gratings, there are also more parameters to optimise the grating for the intended wavelength range. The relationship between efficiency and wavelength is quite homogeneous for VPH gratings, while reflection gratings are optimised for a specific wavelength and their efficiency decreases significantly outside this wavelength.
Got any questions?
Got any questions?
Whether in food production, feed and agriculture or on-harvester applications, Polytec NIR spectrometers deliver reliable real-time results. Our experts will help you identify the ideal setup for your process and application.